TMRE & TMRM Research
Classic Slow Dyes for Measuring Membrane Potential in Cells and Mitochondria
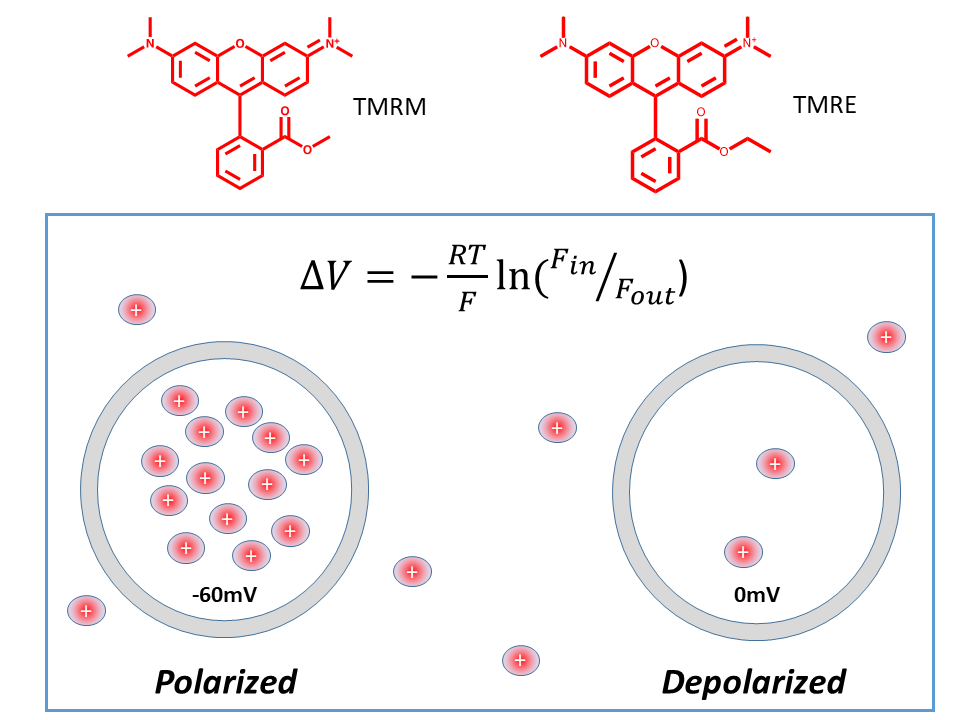
TMRE and TMRM were designed and synthesized in the Loew lab to measure absolute membrane potential via a Nernstian redistribution mechanism (1). The idea is that these permeable highly fluorescent cations will distribute across the membrane according to the Nernst equation. A polarized cell or organelle will, accordingly, have a higher concentration of dye on the inside relative to the outside. At -60mv, a typical resting potential, there will be ~10-fold higher concentration of dye on the inside compared to the outside. Since fluorescence is proportional to concentration, accurately measuring the fluorescence intensity inside the cell using a confocal microscope and comparing it to the fluorescence outside the cell, offers the ability to determine the absolute membrane potential (2). It is also possible to measure changes in membrane potential as a result of signaling activity; however, because the redistribution across the plasma membrane is slow, these dyes are not suitable for fast recording from excitable cells. Additionally, these dyes can be excellent indicators of mitochondrial membrane potential in live cells (2), and with appropriate image analysis can even be used to determine their absolute membrane potential (3-5). Indeed, the use of TMRE and TMRM to assay mitochondrial membrane potential in live cells, qualitatively or quantitatively, has been adopted by many labs using both microscope imaging and cytometry (6-10).
TMRE Makes Mitochondria Light Up in Live Cells
Because mitochondria have typical membrane potentials in the range of -180mV they accumulate very high concentrations of cationic Nernstian dyes like TMRE or TMRM. Shown here is a live NIH 3T3 fibroblast equilibrated with 50nM TMRE. On the left is a maximum intensity projection of a confocal image stack of the entire cell. On the right are 3 optical slices from the region within the rectangle on the full image. With sensitive confocal microscopes, it is possible to use TMRE or TMRM concentrations as low as 5nM to acquire high resolution images.

Citations
1. Ehrenberg, B., V. Montana, M. D. Wei, J. P. Wuskell, and L. M. Loew. 1988. Membrane potential can be determined in individual cells from the nernstian distribution of cationic dyes. Biophysical journal 53:785-794. PMC1330255 PubMed
2. Farkas, D. L., M. D. Wei, P. Febbroriello, J. H. Carson, and L. M. Loew. 1989. Simultaneous imaging of cell and mitochondrial membrane potentials. Biophysical journal 56:1053-1069. PMC1280610 PubMed
3. Loew, L. M., R. A. Tuft, W. Carrington, and F. S. Fay. 1993. Imaging in five dimensions: time-dependent membrane potentials in individual mitochondria. Biophysical journal 65:2396-2407. PMC1225980 PubMed
4. Loew, L. M., W. Carrington, R. A. Tuft, and F. S. Fay. 1994. Physiological cytosolic Ca2+ transients evoke concurrent mitochondrial depolarizations. Proceedings of the National Academy of Sciences of the United States of America 91:12579-12583.45482
5. Fink, C., F. Morgan, and L. M. Loew. 1998. Intracellular fluorescent probe concentrations by confocal microscopy. Biophysical journal 75:1648-1658. PMC1299837 PubMed
6. Ward, M. W., A. C. Rego, B. G. Frenguelli, and D. G. Nicholls. 2000. Mitochondrial membrane potential and glutamate excitotoxicity in cultured cerebellar granule cells. Journal of Neuroscience 20:7208-7219. PMC6772767 PubMed
7. Creed, S., and M. McKenzie. 2019. Measurement of Mitochondrial Membrane Potential with the Fluorescent Dye Tetramethylrhodamine Methyl Ester (TMRM). In Cancer Metabolism: Methods and Protocols. M. Haznadar, editor. Springer New York, New York, NY. 69-76. PubMed
8. Perry, S. W., J. P. Norman, J. Barbieri, E. B. Brown, and H. A. Gelbard. 2011. Mitochondrial membrane potential probes and the proton gradient: a practical usage guide. BioTechniques 50:98-115. PMC3115691 PubMed
9. Chazotte, B. 2011. Labeling Mitochondria with TMRM or TMRE. Cold Spring Harbor Protocols 2011:pdb.prot5641 PubMed
10. Crowley, L. C., M. E. Christensen, and N. J. Waterhouse. 2016. Measuring Mitochondrial Transmembrane Potential by TMRE Staining. Cold Spring Harbor Protocols 2016:pdb.prot087361 PubMed
